Press Release: The potential effect of Plasmacluster Technology on reducing viscous airway mucus that may relieve asthma symptoms using airway stem cell culture - Validated at Columbia University Irving Medical Center, USA


Dr. Munemasa Mori, Principal Investigator and Assistant Professor of Medicine Columbia Center for Human Development, Department of Medicine at Columbia University in New York, U.S., is an expert in respiratory medicine and stem cell research. Dr. Mori and his investigative team have verified the effect of Sharp
Corporation’s Plasmacluster technology on long-term cultured human airway stem cells and confirmed its potential to decrease viscous airway mucus secretion, a problem in asthma patients, suggesting that this technology may help alleviate respiratory tract symptoms related to airway conditions such as asthma.
Airway epithelial cells*2 line the luminal surface of the respiratory tract from the nasal cavity to the lungs. The airway epithelium plays a critical role in mucus clearance by expelling foreign substances through the mucus secretion from secretory cells and the unidirectional beating of motile ciliated cells. The study design
investigated the effects of ions on the respiratory tract. Briefly, human airway tissue stem cells were first induced to differentiate into ciliated and secretory epithelial cells over the course of one month. The cultured cells formed a sheet-like structure and were then exposed to Plasmacluster ions for a maximum of 24 hours.
Dr. Mori’s team did not observe obvious gross morphological changes in the cells or a sign of cell damage.
Instead, quantitative gene expression analysis*3 suggested a decrease in the marker linked with highly-viscous mucus associated with impaired breathing in the respiratory tract of asthma patients and an increase in the marker linked with the smooth secretory protein related to low-viscosity mucus that helps improve breathing. These changes may lead to symptom relief by improving airway mucus secretion, a significant problem in asthma patients. To date, Sharp has demonstrated the effectiveness of Plasmacluster technology in suppressing mite allergens that cause airway problems. Since this test was performed with equipment where no allergens such as dust mites and pollen were present, it suggests that Plasmacluster ions' effects may also contribute to benefit cells directly.
Sharp will further continue to verify the effects on cells and their mechanisms. In addition to measures to counter SARS-CoV-2, Dr. Mori was interested in the overall effects of Plasmacluster ions on the respiratory system. Dr. Mori's team conducted the experiments independently at Columbia University using Plasmacluster technology equipment provided by Sharp.
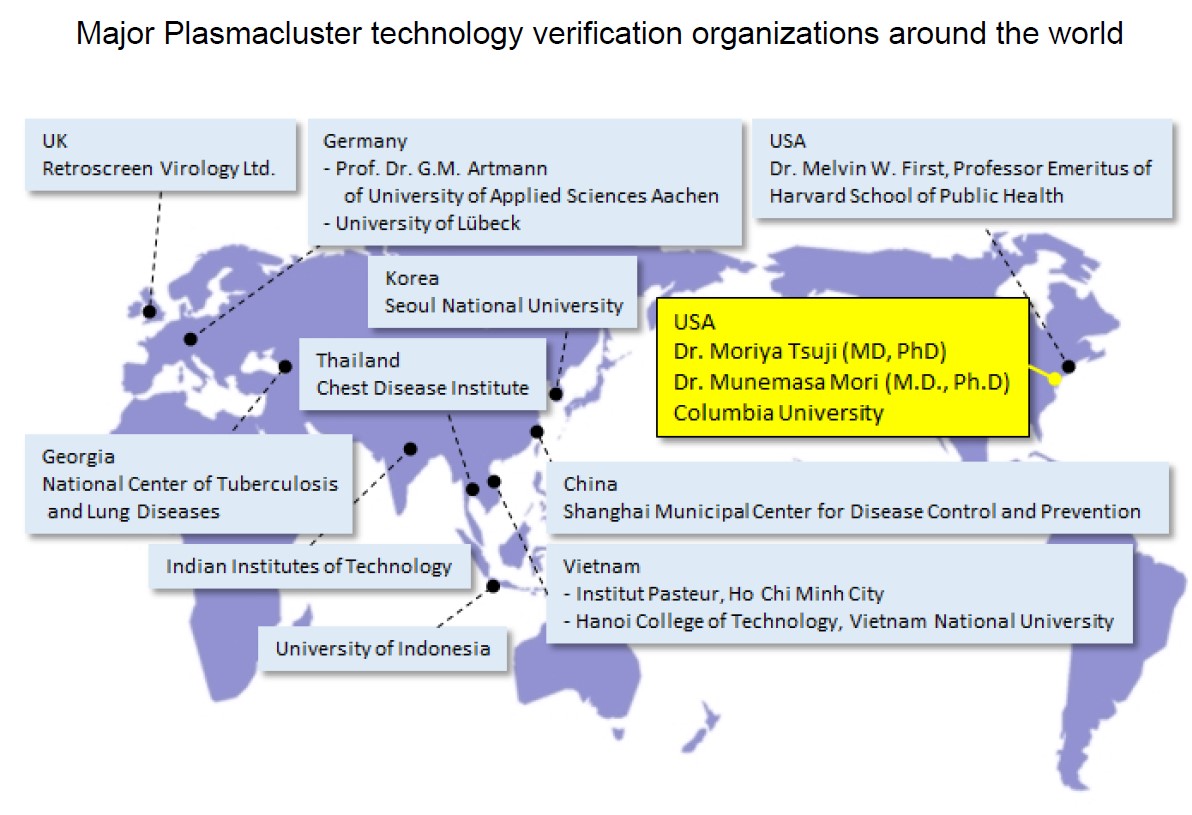
For more than 20 years, Sharp has promoted academic marketing*4 to demonstrate the effectiveness of Plasmacluster technology at independent third-party testing institutions in Japan and overseas. Thus far, numerous independent testing organizations have proven its efficacy in suppressing the activity of harmful substances, including mite allergens, as well as clinical effectiveness in reducing bronchial inflammation levels in children with asthma and prevention and treatment of atopic dermatitis in mice. At the same time, Sharp has continued to verify the safety of Plasmacluster technology over the course of many years.
With the discovery of potential new effects of Plasmacluster technology, which may lead to relieving asthma symptoms in the human respiratory tract, Sharp will continue to demonstrate the effectiveness of Plasmacluster technology actively.
*1 The research results of ion emission type air purification technology suggest the possibility of alleviating asthma symptoms by acting on human airway epithelial cells to alter the mucus markers involved in mucus production. (as of October 13, 2022, based on Sharp findings).
*2 Cells are lining the respiratory tract. They function as an outer layer of the body and are composed of multiple cell types, such as ciliated and mucous cells.
*3 A method of investigating the internal state of cells, an analysis method that examines the functions and reactions of genes.
*4 A marketing method to promote the commercialization of products based on verification of scientific data on the effectiveness of technology in collaboration with leading-edge academic research institutions.
●Plasmacluster and the Plasmacluster logos are registered trademarks of Sharp Corporation.

Figure 1: Test equipment image
Table 1: Microscopy image of cell surface

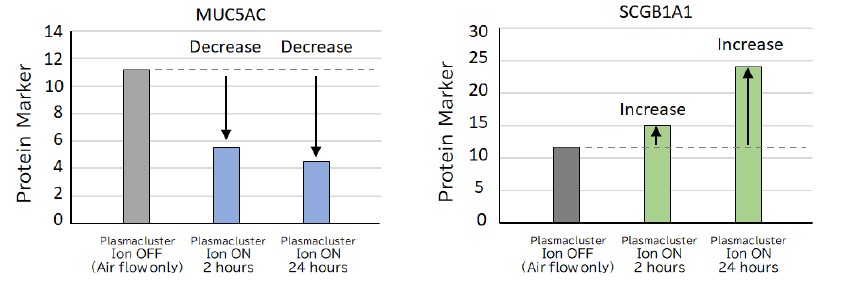
Figure 2: Graph showing the effect of Plasmacluster ions on proteins involved in mucus increase/decrease.
Researcher's Profile: